 |
Gluten: Cereal Grains, Flour & Baked ProductsPhysical-Chemical Properties of Proteins in Baking |
 |
 |
Gluten: Cereal Grains, Flour & Baked ProductsPhysical-Chemical Properties of Proteins in Baking |
 |
Adapted from Wikipedia, the United Nations Food and Agricultural Organization (FAO) and the ChemWiki
Plants have been and will continue to be the primary, 1st order food source for animals, and plant seeds being less perishable than much of the other parts of plants are very important in storing food and energy. Grains and cereals are at the center of human's food, and in our global economy the developed world has access to most all of the grains grown domestically and in-the-wild throughout our bio-sphere. The current major dietary grains, which are in the grass family Gramineae or Poaceae, are wheat, which is shown in the image above left, rice and corn (maize). They can be traced respectively to Europe, Asia, and meso-America (Mexico, Belize, Guatemala, El Salvador, Honduras, Nicaragua, and Costa Rica). There are many other grasses such as sorghum, millet, rye, barley, oats, quinoa, and others that thrive throughout the world depending on the climate and growing conditions. There are over 11,000 grass species. They are found to grow on all of the continents including Antarctica.
Wheat Seed:
A wheat grain consists of three main parts: the endosperm, the bran, which is mostly wall tissue and wheat germ. The germ is rich in oil and it contains lots of vitamins. The endosperm is where the starch is stored. It also encapsulates protein that transforms into gluten during kneading.
The seed wall, which has a large amount of relatively more difficult to digest polysaccharides, is made of several layers; The aleuron layer, which is also rich in proteins and vitamins, separates the endosperm from the adjoining layers.
Wheat is ground or milled to separate the endosperm from the wall layers and the germ. During the grinding the endosperm is reduced to what we know as flour, after grinding the wall layers and germ make up the bran portion.
Once seeds are collected for food, they need to be processed to make them more edible by chemically and mechanically treating their hard outer coat making them easier to digest, which allows the biochemical machinery in our bodies to process them into useful molecules and to produce energy, which we use to do work, both manual and intellectual, while maintaining (homeostasis) and replenishing our bodies (eg. red blood cell replacement, where about 100 million new red blood cells are being formed in our body every minute! ) http://book.bionumbers.org/how-quickly-do-different-cells-in-the-body-replace-themselves/
Baking “breads” from grains is arguably among the oldest, most practiced and developed form of cooking with an enormous number of different types of grain-based finished products: unleavened breads, leavened breads, sourdough breads, flatbreads, cornbreads, pita, lavash, naan, focaccia, tortillas, bisquits, crispbreads, damper, bagels, waffles, pancakes, and on and on. (https://en.wikipedia.org/wiki/List_of_breads)
Food science and nutrition focus on three broad categories of bio-molecules: proteins, saccharides (starch & sugars) and fats (lipids). The thermodynamic properties of these three types of molecules are very important to many of us, that is, being concerned with “How many Calories does it have?”. Aside from being self-aware of the Calories in what we eat and the consequential health effects, it is important to recognize that all three categories are essential and interrelated. Yet, they tend to be labeled as “good” for you or “bad” for you, eg. antioxidants or trans-fats. All three types of the bio-molecules are found in grains, but their relative quantities are not distributed evenly, and they have complex interactions when their flour, which is produced from grinding the grain, is processed and cooked.
Compositionally, cereal grains consist of 12-14 percent water, 65-75 percent carbohydrates, 2-6 percent lipids and 7-12 percent protein. They are quite similar in gross composition being low in protein and high in carbohydrates. The proteins are found enclosed between the starch grains. Wheat proteins can be classified into four general classes, which are distinguished by their solubility:
a. albumin : dissolves in water
b. globulin : dissolves in salt solutions
c. gliadin : dissolves in 70 % alcohol
d. glutenin : dissolves in dilute acids or basesThe first two form about 20% of the total amount of protein, while the glutenin and gliadin contribute the remaining 80 %.
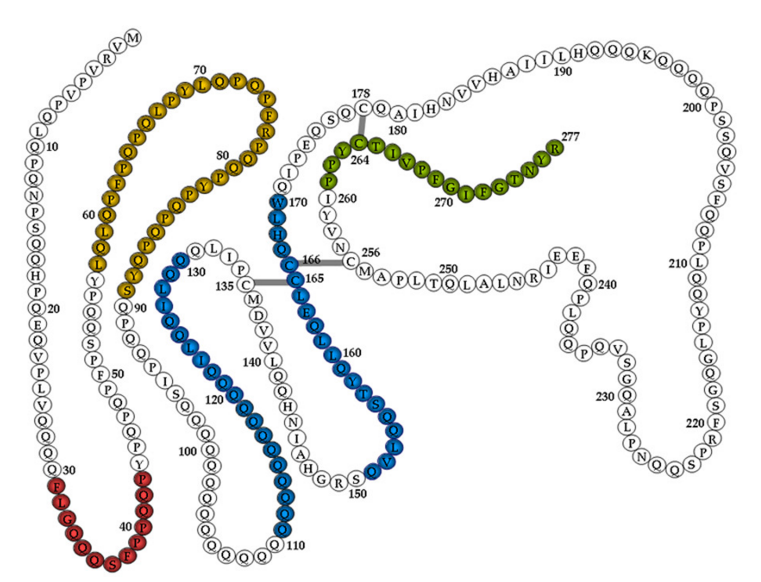
In baking, a very important protein is gluten, which forms from two other smaller proteins found in certain cereal grains, gliadin [a tetrapeptide portion of its structure and chemical information is shown above (http://molview.org)] and glutenin. Researchers have found that alpha-gliadin has 266 amino acids, with over 60 glutamines and over 30 prolines in its overall primary structure. This is very unusual since most proteins have a fairly random scattering of amino acids. For wheat cultivars, two amino acids, glutamine, which ranges from 36% up to 45% of the total amino acids and proline from 17% to 23%, comprise more than half of the amino acids. There are 9 essential amino acids: histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine. These are produced only in plant cells, are of great importance for health and nutrition in animals and humans, who cannot sythesize these amino acids themselves, and must be acquired through diet. Simple proteins are anabolic building blocks in human metabolism, but in plants they act as storage materials in the seeds. Glutenin is a polypeptide formed from essential amino acids, but in contrast gliadins lack any essential amino acids. The two are present in varying amounts in many flours, and when mixed with water and mechanically processed (kneading) the two form new sulfur-sulfur bonds that cross-link the gliadin and glutenin protein chains together to from the larger protein, gluten. This process is similar to hair stylists forming “permanent” waves, where chemically treating hair and rearranging sulfur-sulfur bonds produces curls. (http://evoice.ewha.ac.kr/news/articleView.html?idxno=517)
The newly formed gluten molecules form a physical-chemical 3-dimensional network that can trap and hold carbon dioxide gas, which is a product of fermentation in yeast breads. It is this gluten network that causes bread to rise by trapping the CO2 gases, which are produced by the enzymatic reaction of yeast that acts as a catalyst.
Small changes in baking recipes, such as which type of flour is used, what other ingredients are added, the pH and quality of the water used, and the mixing method can have a large affect on gluten development, the properties of the dough produced, and the quality of the baked goods that come out of the oven.
Beware, humans need to be cautious in eating bread and pastry. Eating gluten can trigger disorders in susceptible people that include celiac disease (CD), non-celiac gluten sensitivity (NCGS), gluten ataxia (GA), dermatitis herpetiformis (DH) and wheat allergy (WA). The most common of these, celiac or celiac-sprue disease, is described in the NY Times Health Guide (http://www.nytimes.com/health/guides/disease/celiac-disease-sprue/overview.html).
Celiac disease is a condition that creates inflammation and damages the lining of the small intestine. This prevents absorbing components of food that are important for staying healthy. The damage is due to a reaction to eating gluten, which is found in wheat, barley, rye, and possibly oats.
The exact cause of celiac disease is unknown. The lining of the intestines is covered by villi, which help absorb nutrients. When people with celiac disease eat foods or use products that contain gluten, their immune system reacts by damaging these villi. This damage affects the ability to absorb nutrients properly. A person becomes malnourished, no matter how much food he or she eats.
The disease can develop at any point in life, from infancy to late adulthood. People who have a family member with celiac disease are at greater risk for developing the disease. The disorder is most common in Caucasians and persons of European ancestry. Women are affected more often than men.
The symptoms of celiac disease can be very different from person to person and cover a wide range of seemingly opposite pathologies, or be asymptomatic. This is part of the reason why diagnoses cannot always made right away. For example, one person may have constipation, a second may have diarrhea, and a third may have no problem with stools.
Gastrointestinal symptoms may include one or more of the following:
- Abdominal pain, bloating, gas, or indigestion
- Constipation Decreased appetite (may also be increased or unchanged)
- Diarrhea, either constant or off and on
- Lactose intolerance (common when the person is diagnosed, usually goes away after treatment)
- Nausea and vomiting Stools that are foul smelling, oily, or and stick to the toilet when flushed
- Unexplained weight loss (although people can be overweight or of normal weight)
Globally, celiac disease affects between 1 in 100 and 1 in 170 people. Rates, however, vary between different regions from as few as 1 in 300 to as many as 1 in 40. It is most common in people of European ancestry; People of Indian ancestry seem to have a similar risk as Europeans; People who are of African, Japanese and Chinese descent are rarely diagnosed; this global distribution reflects inherent genetic risk factors in each population.
Gluten-free diets have been endorsed by Miley Cyrus and other celebrities, and are used by non-celiac diseased people, who believe that the diet can improve energy and health. A 2011 best selling book, Wheat Belly, refers to wheat as a "chronic poison" and leads the reader to believe that removing gluten from one’s diet will help them lose weight, improve their health, and generally feel better. The author’s blog (http://www.wheatbellyblog.com/) is advertising the latest release in a series of similar books. “Check it out — Dr. Davis newest book, Wheat Belly 10-Day Grain Detox, is in stores now!”
From the early days of a few small health food stores selling gluten free foods to the present day, where major supermarkets have gluten free sections and restaurants offer gluten free menu choices, a growing number of followers have adopted a gluten free diet. One survey indicates that as many as 100 million people will buy gluten free products in 2016.
The Harvard University health blog (http://www.health.harvard.edu/blog/going-gluten-free-just-because-heres-what-you-need-to-know-201302205916) advises that “… it’s become hip to go gluten-free. Based on little or no evidence other than testimonials…”
WebMD (http://www.webmd.com/diet/20120220/gluten-sensitivity-fact-or-fad) advises ….’that common sense must prevail to “prevent a gluten preoccupation from evolving into the conviction that gluten is toxic for most of the population.”
Scientific American published an article citing the downside of gluten free diets: “Most People Shouldn't Eat Gluten-Free” (http://www.scientificamerican.com/article/most-people-shouldnt-eat-gluten-free/).
Like any chemical reaction, the reactants, gliadin and glutenin, must physically come in contact on a molecular level for a reaction to occur and the product, gluten, to form. Considering the effects of kneading, dough formation is consistent with the collision theory of chemical kinetics. The two smaller protein reactants do not come together to create gluten until they are placed in water, which assists in the molecular interaction, and then are mechanically worked together by kneading. Once water is added and gluten forms, the new, strong covalent sulfur-sulfur bonds produce a new protein structure, which provides important properties to the dough. But, there needs to be balance. Too many disulfide/ dithio (S-S) bonds tend to make the dough stiff and too many “free” thiol groups (SH) make the dough flabby, which is demonstrated by adding extra thiol groups in the form of cysteine to the dough. This suggests that there should be very little difference in the relative amounts of the two functions, whichever of these may be the “limiting function” much like a “limiting reactant”.
Without a balanced number of covalent bonds in the gluten molecules, the dough lacks the physical-chemical properties that produce quality baked goods.
Refer to: https://bondingwithfood.wordpress.com/
Gluten-free dough is not able to bind as well as dough with gluten or to bind at all. To overcome this problem, gluten-free flours use several different additives such as xanthan or guar gum, gelatin, sweet whey, eggs, raw buckwheat flour, chia or flaxseed slurries, which help bring back some of the stickiness lost from the missing gluten. Types of flours and respective gluten content: http://whatscookingamerica.net/Bread/FlourTypes.htm
Test the science. Visit the Web site: “Cookie Science 2: Baking a testable hypothesis”:
https://student.societyforscience.org/blog/eureka-lab/cookie-science-2-baking-testable-hypothesisChoose two of the 6 home experiment(s), one with gluten and one without. There are 3 cookie recipes with gluten and 3 cookie recipes without gluten to choose from.
Experimental Procedure:
- View:
https://www.youtube.com/watch?v=H2YBXcDUMTk
Your working group has been assigned three different types of flour.
Before beginning the experiment, complete the background reading and view the YouTube video using the above link.
Procedure:
- Weigh out ~100. grams of each of your flours in individual weighing boats (plastic dishes) using one of the in-lab balances (NOT an analytical balance). As viewed in the video, use the lab bench as your work space after wiping it with water and drying it with a paper towel to mix the flour with water and to knead the dough, Be sure to keep track of which flour is which, and have a small amount of extra flour on-hand for dusting the work space and the dough, as you may need.
- Slowly add about 30-40 mL of water to each flour until it has a consistency that allows it to be handled and kneaded, adding water as needed. The amount of water added does not need to be recorded. It will vary with each type of flour.
- Knead each dough by hand for ~5 minutes, until the gluten is fully developed producing a soft, rubbery ball of dough. Let the dough ball sit (rest) for ~10 minutes.
- Place the dough balls in separate containers or bowls and fill the containers with cool water. Let the gluten balls soak in the water for ~10 minutes. Tear apart each dough by hand while submerged in water. You will notice that the water turns milky as it washes away the starch in the dough. Occasionally pour out the cloudy water as it collects in the bottom of the container. Slowly, the dough balls will become a gummy, slimy network of gluten strands, which you will not be able to see with the naked eye. If the dough is no longer a ball, collect all of the bits and pieces and clump together.
- Remove the dough balls (or pieces) and slowy run cold water over each of them. Be careful not to let the dough disintegrate; try cupping your hands around the ball and squeezing gently to remove any starch that is left behind. Continue this process until the water squeezed from gluten ball is almost clear. The ball should become smaller and more stretchy.
- Drain off and squeeze the gluten balls (or clumped pieces) to remove as much excess water as possible. (Rye, rice, corn, and other flour do not form gluten balls.
Pat each dry.
- Allow them to relax (rest) for a minimum of fifteen minutes before evaluating them. This allows time for the gluten network to recover from the washing process.
Evaluation:
- Weigh each on an in-lab balance (NOT an analytical balance) and record the results. Rank the relative size of the gluten balls from largest (1) to smallest (3) based on their volume and record the rank in the table.
Gently pull each ball between your hands. Rotate the ball while pulling, so that you are pulling it in all directions. Based on these tests rank the following properties from highest (1) to lowest (3) in the table.
- Strength (tenacity): The harder it is to stretch, the stronger the gluten. If the gluten ball falls apart and is not cohesive enough to stretch, record (NS), that is, it does not stretch.
- Elasticity/ springiness (how well the gluten ball springs back when pressed or stretched)
- Calculate the percent gluten in flour as follows, and record results in the table.
% Gluten = (100 x Weight of Gluten Ball) ÷ (3 x Weight of Flour)
The % calculation is based on the assumption that gluten absorbs two times its weight in water, meaning that every ounce (~30 grams) of the gluten ball is (~10 grams) gluten. This calculation also assumes that the gluten ball is only gluten. In fact, some lipids, starch, and other non-protein materials are trapped in the gluten.
(Report form )
Post Lab Questions:
- How does the calculated percent of gluten compare to the dough’s strength & elasticity?
- What is the % of hydrophilic amino acids in the total number of amino acids of the following gliadin string? Show your calculation.
FPQPQPFPPQ LPYPQPPPFS PQQPYPQPQP QYPQPQQPIS QQQAQQQQQQ
- What does kneading do to the property of dough? Can too much kneading affect the quality of the dough? Briefly explain your answers and reasoning.
- Which level(s) of protein structure are significant in explaining the general elasticity of dough? Circle all that apply. 1o, 2o, 3o, 4o Briefly explain the reason(s) for your choice(s).
References:
The four structural levels of proteins are:
• 1o (primary): The sequence of amino acids, eg. ARDV: Ala.Arg.Asp.Val.
H2N- on the left and on the right -COOH• 2o (secondary): Structures which include, folds, turns, alpha-helices, and beta-sheets held in place by hydrogen bonds.
• 3o (tertiary): 3-d arrangement of all atoms in a single polypeptide chain.
• 4o (quaternary): Arrangement of all polypeptide chains into a functional protein, eg. hemoglobin with 4 strands: 2 pairs of 2 chains, one "alpha" and one "beta"