Pre lab Questions
1) Refer to the gas chromatograph (below) of a mixture of Compounds
A and B:
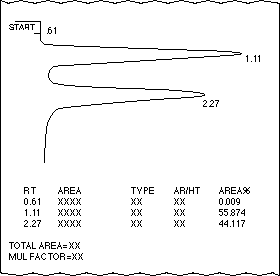
-
Which compound has the longer retention time?
-
Which compound is present in a larger amount?
-
Which compound has the lower boiling point?
-
What would happen to the retention times of compounds A and B if the column
temperature were raised?
2) Consider the following compounds:
| compound |
boiling point
|
| methyl cyclohexane |
101
|
| pentane |
36
|
| octane |
126
|
| 2,3-dimethyl octane |
165
|
| heptane |
98
|
If the compounds in the table above are run on an OV-101 GC column,
rank the compounds in increasing order of retention times.
3) Consider the following compounds:
| compound |
boiling point |
| propionic acid |
141
|
| 2-hexanol |
140
|
| isoamyl acetate |
142
|
| 3,4-dimethylheptane |
140
|
If they are run on an DEGS GC column, rank the compounds in decreasing
order of retention times.
4) You think that you injected your sample into the GC and then wait several
minutes, but you see no peak. What should you do? (Ask Dr. R. is an incorrect
response.)
5) State the stationary and moving phases in each type of chromatography
below:
-
thin layer (TLC)
-
gas (GC)
6) Rank the following compounds in terms of their expected GC retention
times on OV-101, a non-polar stationary phase, and their relative rank
by Rf values on a silica gel TLC plate that is eluted with a
non-polar solvent.



